|
Listen to this article  |
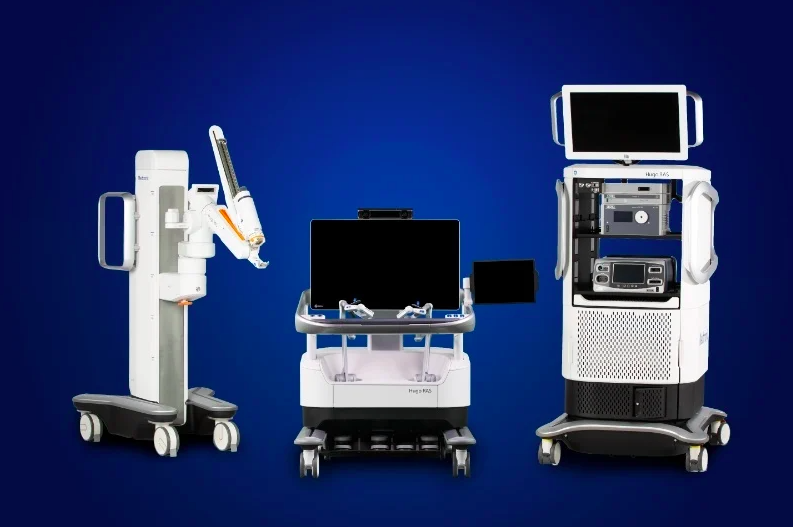
Hugo includes wristed instruments and 3D visualization to help surgeons perform minimally-invasive surgeries. | Credit: Medtronic
Medtronic received CE mark approval in Europe for its Hugo robotic-assisted surgery system. Fridley, Minnesota-based Medtronic’s CE mark covers urologic and gynecologic procedures, which make up approximately half of all robotic procedures performed today, according to the company.
Hugo, which in May 2021 garnered FDA investigational device exemption, offers a modular, multi-quadrant platform designed for a broad range of surgical procedures with wristed instruments, 3D visualization and surgical video capture option. In June, Medtronic announced the first clinical procedure with Hugo in Chile.
“This day has been a long time coming, not just for Medtronic, but for the surgeons and hospital leaders who have partnered with us on this journey to bring the benefits of robotic-assisted surgery to more patients around the world. We know our solution is more meaningful because of their insights,” said Megan Rosengarten, president of Medtronic’s surgical robotics business. “With the Hugo RAS system in our European customers’ hands and our dedicated team by their side, together we will redefine what is possible in robotic-assisted surgery.”
Multiple hospitals will be among the first in Europe to join Medtronic’s Partners in Possibility Program to use the Hugo system and participate in a global patient registry. Clinicians from the hospitals will participate in hands-on training at Medtronic Surgical Robotics Experience Centers, including two sites in Ghent, Belgium, and Strasbourg, France.
“Robotics and artificial intelligence are the undeniable future of healthcare, with incredible potential to not only advance patient care, but increase access to these benefits,” added Medtronic EVP & president of the EMEA region Rob ten Hoedt. “The Hugo RAS system builds on our leadership in minimally invasive surgery, and we’re thrilled to provide hospitals across Europe a robotic-assisted surgery system that is thoughtfully designed to meet their needs today and tomorrow.
“We’ve had strong interest from leading surgical centers across Europe and expect to move quickly with multiple installations in several countries.”
Editor’s Note: This article was first published by sister publication Mass Device.
Credit: Source link


Comments are closed.