|
Listen to this article  |
Microbot Medical Inc. has successfully completed a pre-clinical study of its Liberty robotic system for endovascular surgery. The Braintree, Mass.-based company also announced that it is working with Corewell Health Forge to enable teleoperation of Liberty for remote procedures.
Microbot Medical officials said in a release that remote operations with Liberty could help democratize endovascular interventional procedures. Unlike other robot-assisted surgical systems, Liberty is designed to eliminate the need for large, cumbersome, and expensive capital equipment, they said.
 Submit your nominations for innovation awards in the 2024 RBR50 awards.
Submit your nominations for innovation awards in the 2024 RBR50 awards.
Study a step toward FDA approval of Liberty
The pre-clinical study is necessary step toward Microbot Medical’s investigational device exemption (IDE) submission to the U.S. Food and Drug Administration (FDA). It took place under rigorous FDA guidelines for good laboratory practice (GLP) and involved pigs.
Three leading interventional radiologists conducted the study, using Liberty to perform a total of 96 robotic navigations. They examined and evaluated target vessels with surrounding tissue microscopically after subjecting them to procedures using a range of commercially available intravascular catheterization devices controlled and manipulated with the company‘s system.
“I am very pleased with the positive outcomes of the histopathology report and the completion of the GLP study,” stated Juan Diaz Cartelle, chief medical officer of Microbot Medical. “This gives us confidence to move forward to the next stage of human clinical study.”
“Today’s announcement marks another important milestone for the company as we continue our transition from R&D and pre-clinical phase into the clinical, regulatory, and pre-commercial phase,” added Harel Gadot, CEO. “We expect to submit our IDE application to the FDA soon and commence our pivotal human clinical study, completing our transition to a clinically stage company.”
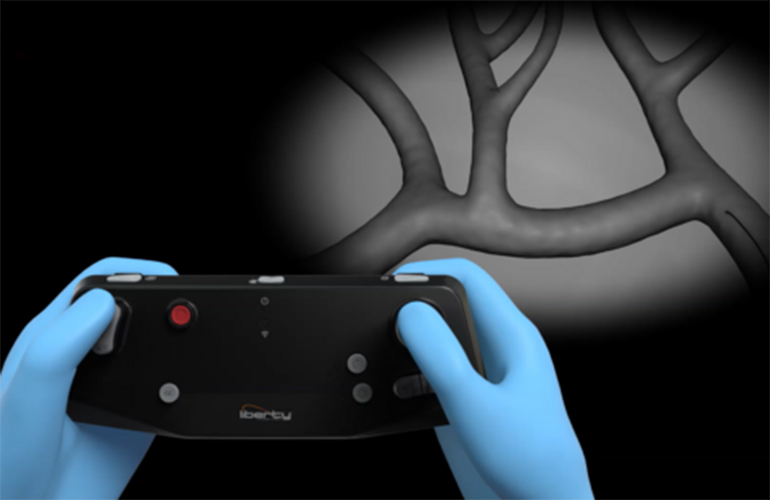
The Liberty robotic surgical system. Source: Microbot Medical
Microbot, Corewell to jointly evaluate surgical robot
Microbot Medical said its collaboration with Corewell Health, which will take place in multiple phases, will enable telerobotics between remote centers. The agreement includes the mutual evaluation of the Liberty system for remote procedures. Ryan Madder, M.D., an expert in interventional cardiology, will lead that effort.
“Being able to connect between remote sites, especially at rural areas, has been a passion of mine for years,” said Dr. Madder in a release. He is also section chief for interventional cardiology and director of Cardiac Cath Lab at Corewell Health in Grand Rapids, Mich.
“I’m excited about the potential this collaboration can bring to benefit many patients in the USA and across the world,” he added.
Corewell Health is a not-for-profit health system with more than 60,000 employees, including over 11,500 physicians and advanced practice providers and more than 15,000 nurses. It said it provides care and services in 21 hospitals, over 300 outpatient locations, and several post-acute facilities.
“We are excited for the opportunity to collaborate with Corewell Health and Dr. Ryan Madder, innovators in the field of robotics and healthcare,” said Gadot. “Working together with a health system such as Corewell … to enhance the telerobotic feature of the LIBERTY System fits well with our mission to enable accessibility to any user, anywhere across the world for any endovascular procedure.”
“We continue to demonstrate that the interest shown by leading medical centers in our Liberty system is a testament to its potential impact in the endovascular interventional space,” he said.
Editor’s note: This article was syndicated from MassDevice, a sibling site to The Robot Report.
Credit: Source link


Comments are closed.